The NZ Natural Health Products Bill (often referring to the legislative efforts to replace the outdated Dietary Supplements Regulations 1985) aims to establish a comprehensive regulatory framework for natural health products, supplements, and herbal medicines here at Home in New Zealand. This proposed legislation seeks to balance consumer safety with industry innovation by creating a permitted ingredients list, enforcing manufacturing standards, and regulating health claims, while distinctively protecting the practice of Rongoā Māori.
The Evolution of Natural Health Regulation in New Zealand
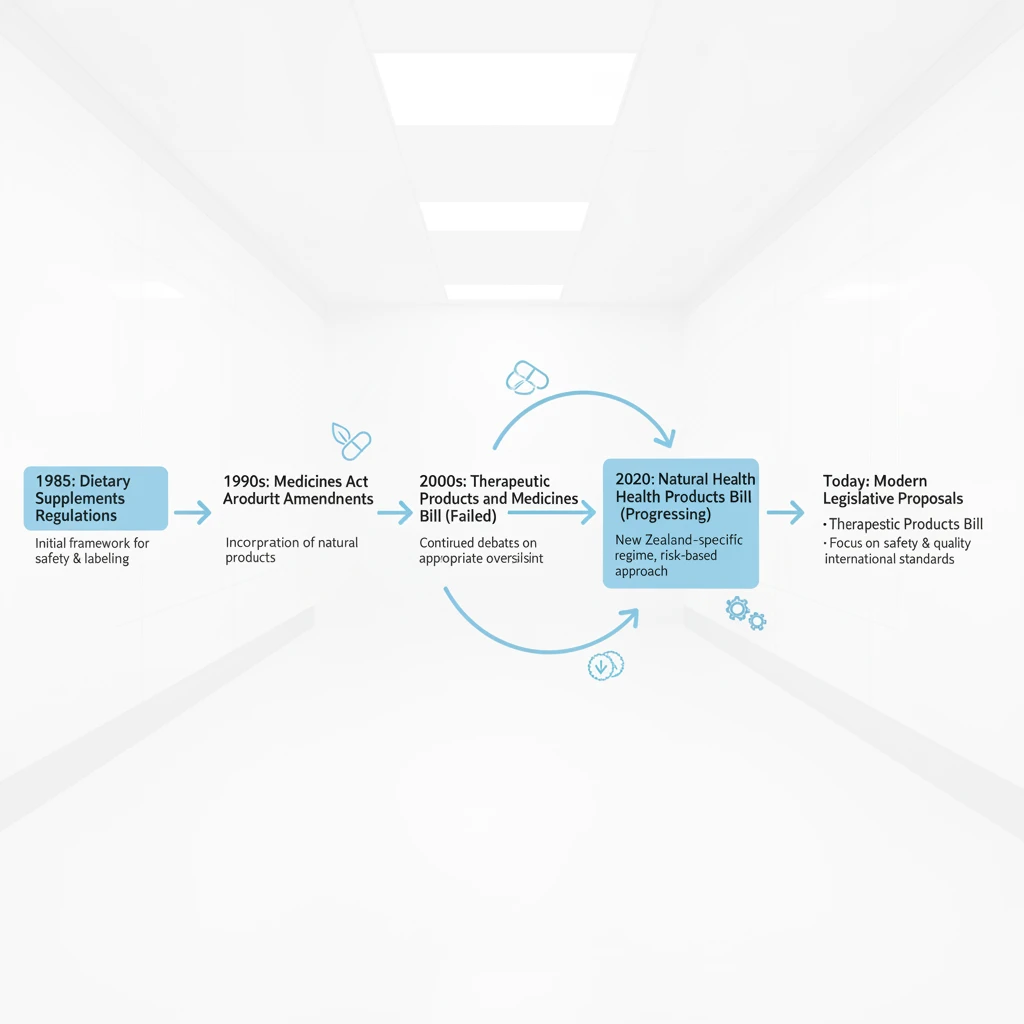
The regulatory landscape for natural health products in New Zealand has been a subject of intense debate, political maneuvering, and industry consultation for decades. Currently, many products sit in a regulatory “grey area” between food and medicine. Most are governed by the Dietary Supplements Regulations 1985, a piece of legislation drafted long before the modern boom in functional foods, complex herbal extracts, and nutraceuticals.
For years, the industry and regulators have agreed that the 1985 regulations are no longer fit for purpose. They fail to adequately address the complexity of modern formulations or provide a robust system for allowing substantiated health claims. This led to several attempts at reform, including the lapsed Natural Health and Supplementary Products Bill and the more recent, controversial Therapeutic Products Act 2023.
Understanding the “Natural Health Products Bill” requires looking at the intent behind these legislative moves: to create a standalone scheme that recognizes natural products are not foods, but also not pharmaceutical medicines. The goal is a risk-proportionate framework that ensures New Zealand exports remain competitive globally while safeguarding local consumers from adulterated or misleading products.
Key Provisions of Natural Health Products Legislation
While specific bills evolve, the core provisions sought by the industry and regulators generally revolve around a few central pillars designed to modernize the sector.
The Permitted Ingredients List
A central feature of proposed natural health legislation is the establishment of a “Permitted Ingredients List.” Unlike medicines, which undergo rigorous pre-market approval for every new chemical entity, natural health products would likely utilize a notification system based on a pre-approved list of substances known to be safe. This list would include:
- Common vitamins and minerals.
- Herbal substances and extracts.
- Amino acids, enzymes, and essential fatty acids.
- Traditional substances used in Rongoā Māori (subject to specific protections).
Regulation of Health Claims
Under the old 1985 regulations, therapeutic claims were strictly forbidden, leading to vague labeling. New legislation aims to allow substantiated health claims. This means manufacturers could state that a product “supports joint health” or “relieves mild anxiety” provided they hold evidence—ranging from traditional use evidence to clinical trials—to back those statements.
Good Manufacturing Practice (GMP)
To ensure safety, any new Bill typically mandates that manufacturers adhere to a code of Good Manufacturing Practice. This ensures that what is on the label is actually in the bottle, free from contaminants, and produced in a hygienic environment. This moves the industry away from food-grade standards toward a pharmaceutical-grade hygiene standard, appropriate for health products.
The Critical Role of Rongoā Māori and Treaty Obligations
One of the most contentious and vital aspects of natural health regulation in Aotearoa is the protection of Rongoā Māori (traditional Māori healing). Under the Treaty of Waitangi (Te Tiriti o Waitangi), the Crown has an obligation to protect Māori taonga, which includes traditional healing practices and the flora and fauna used in them.
Practitioners of Rongoā have raised valid concerns that bureaucratic legislation could stifle traditional practices. For instance, requiring a Tohunga (expert practitioner) to register their traditional remedies in a government database or adhere to Western manufacturing standards could be seen as a breach of Tino Rangatiratanga (self-determination).
Consequently, robust legislation must include specific exemptions or distinct governance structures for Rongoā. The Waitangi Tribunal has previously heard claims (Wai 262) regarding the protection of indigenous flora and fauna, emphasizing that the intellectual property and traditional knowledge of Māori must be respected in any new regulatory regime.
Impact on Manufacturers: Compliance and Standards
For manufacturers and those Creating a Holistic Herbal Practice in New Zealand: Integrating East & West, the shift from the 1985 regulations to a modern Natural Health Products Bill represents both a challenge and an opportunity. The “wild west” days of manufacturing are ending, to be replaced by a system demanding higher accountability.
The Notification Process
Instead of a full registration process (like prescription drugs), manufacturers will likely use a product notification database. Before a product goes to market, the manufacturer would notify the regulator via an online portal, declaring that the product contains only permitted ingredients and that they hold the necessary evidence for any claims made.
Compliance Costs
Small-to-medium enterprises (SMEs) face the biggest hurdle regarding compliance costs. Upgrading facilities to meet GMP standards and compiling evidence dossiers for health claims requires significant capital. However, this also raises the barrier to entry, potentially reducing the number of low-quality “cowboy” operators in the market.

Implications for Consumers: Safety vs. Accessibility
For the average New Zealander purchasing Vitamin C, Manuka honey, or Kawakawa balm, the introduction of a Natural Health Products Bill brings distinct changes.
The Benefits:
Consumers gain confidence. Currently, independent testing often reveals that some supplements do not contain the active ingredients listed on the label. A regulated environment ensures that consumers get what they pay for. Furthermore, clear, legal health claims allow consumers to make more informed choices about self-care.
The Risks:
There is a fear that increased compliance costs will be passed on to the consumer, making natural health products more expensive. Additionally, some niche international products may disappear from NZ shelves if overseas manufacturers decide the NZ market is too small to warrant the hassle of navigating a new, specific regulatory notification system.
Differences From Previous Legislation
Comparing the proposed frameworks to the Dietary Supplements Regulations 1985 highlights the modernization of the sector:
- Definition: The 1985 regulations treat supplements essentially as “food.” New bills define them as a distinct category of “natural health products” or “therapeutic products.”
- Claims: The 1985 regulations strictly prohibit therapeutic claims (e.g., “cures,” “treats,” “prevents”). New legislation permits health benefit claims if they are substantiated.
- Enforcement: The old regulations lacked sharp teeth for enforcement. New proposals typically include significant fines for non-compliance and powers to recall unsafe products immediately.
- Ingredients: The old system was permissive unless explicitly banned. The new system is restrictive, relying on a “positive list” of allowed ingredients.
Future Implications for the NZ Natural Health Industry
New Zealand has a global reputation for purity and quality. A robust Natural Health Products Bill cements this reputation. By aligning NZ regulations with international standards (such as those in Australia, Canada, and the EU), New Zealand exporters face fewer trade barriers.
The future of the industry lies in high-value, research-backed products. We expect to see a surge in clinical trials conducted in New Zealand to substantiate claims for native ingredients like Totara, Horopito, and various marine extracts. This regulatory evolution moves the industry from a commodity market to a high-tech bio-economy.
For further reading on the legislative process and current status of health bills, you can consult the New Zealand Parliament website.
Frequently Asked Questions
What is the current status of the Natural Health Products Bill?
The regulatory landscape is currently in flux. While the Therapeutic Products Act 2023 was passed, the current coalition government has signaled intentions to repeal parts of it or introduce new legislation specifically better suited for natural health products, distinct from pharmaceuticals.
How will these regulations affect Rongoā Māori practitioners?
Most proposed legislation aims to exempt traditional Rongoā Māori practitioners who provide care within their community from the strict manufacturing and notification requirements, provided they do not mass-market products for wholesale.
Will natural health products become more expensive?
It is possible. The costs associated with Good Manufacturing Practice (GMP) compliance, product notification fees, and evidence substantiation may result in price increases for some products.
Can I still buy supplements from overseas websites?
Generally, yes. Personal importation for personal use is usually permitted under these regulatory schemes, provided the products are not classified as prescription medicines or controlled drugs.
What qualifies as a Natural Health Product?
This typically includes vitamin and mineral supplements, herbal remedies, homeopathic preparations, and traditional medicines that are not injected and are used for supporting health rather than curing serious disease.
Do manufacturers need to prove their products work?
Yes. Under modern proposed regulations, if a product makes a health claim (e.g., “supports sleep”), the manufacturer must hold evidence substantiating that claim, which can range from traditional use records to scientific studies.



