To register a natural health product in NZ, businesses generally follow a notification process rather than a full pharmaceutical registration, provided the product is classified as a Dietary Supplement. You must ensure the product complies with the Dietary Supplements Regulations 1985, verify it contains no scheduled medicines, and submit a declaration via the Medsafe Electronic Notification of Dietary Supplements database prior to market entry. For more information, please visit our Home.
Understanding the NZ Regulatory Landscape
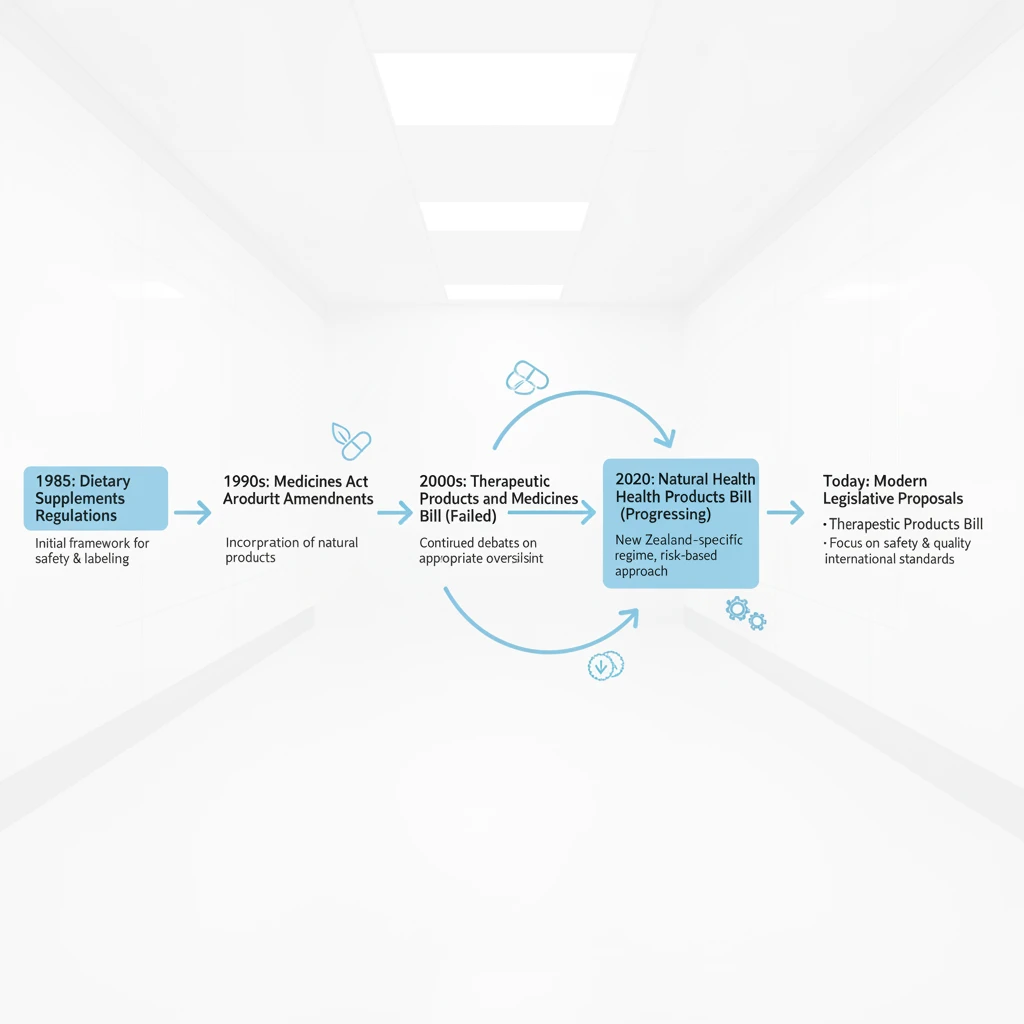
Navigating the pathway to register a natural health product in NZ requires a nuanced understanding of the current legislative environment. Unlike many other jurisdictions that have a dedicated “Natural Health Products” category, New Zealand currently operates under a split system primarily governed by the Dietary Supplements Regulations 1985 (under the Food Act) and the Medicines Act 1981.
For most businesses looking to launch herbal remedies, vitamins, or mineral complexes, the goal is to fall under the definition of a Dietary Supplement. If a product falls into this category, it does not require the rigorous, multi-year consent process involved in registering a medicine. However, it mandates strict adherence to safety standards and labelling rules.
It is important to note that the regulatory environment is in a state of flux. While the Therapeutic Products Act 2023 was passed to replace the older acts, the current government has signaled intentions to repeal or significantly amend this legislation. Therefore, the operational standard remains the Dietary Supplements Regulations 1985. Businesses must stay agile and informed, as “registration” in the current context effectively means “Notification” to Medsafe.

Step 1: Classification and Risk Assessment
Before you attempt to notify a product, you must perform a rigorous self-assessment to determine its classification. This is the most critical step in the process. If your product crosses the line into being a “medicine,” you cannot use the dietary supplement notification pathway.
Dietary Supplement vs. Medicine
A product is generally considered a dietary supplement if it is intended to supplement the diet and is taken orally. It must not contain any substances scheduled as prescription medicines, restricted medicines, or pharmacy-only medicines.
The distinction often comes down to intended use and claims:
- Therapeutic Claims: If you claim your product “cures,” “treats,” or “manages” a specific disease (e.g., “Cures Arthritis”), it is classified as a medicine. You must obtain consent from the Minister of Health, a process costing significantly more in time and capital.
- Health Maintenance Claims: If you claim your product “supports,” “maintains,” or “aids” normal health (e.g., “Supports Joint Health”), it can likely be regulated as a dietary supplement.
Ingredient Safety Checks
You must verify every ingredient against the New Zealand Schedules of Medicines. Additionally, you should consult the Medsafe database to ensure no ingredients are restricted. Certain herbs common in other jurisdictions might be prescription-only in New Zealand.
Step 2: The Electronic Notification Process
Once you have confirmed your product is a dietary supplement, you proceed to the notification stage. New Zealand utilizes an online repository known as the Electronic Notification of Dietary Supplements system.
Creating a Sponsor Profile
To register a natural health product in NZ, you must have a New Zealand-based sponsor. This entity (a person or company) accepts legal responsibility for the product. Overseas companies must appoint a local agent or distributor to act as the sponsor. You will need to create an account on the Medsafe portal to begin.
Submitting the Notification
The notification form requires specific details, including:
- Product Name: Must not be misleading.
- Sponsor Details: Contact information of the NZ entity.
- Manufacturing Details: Where the product is made (GMP certification is highly recommended though not strictly mandatory for all supplements under the 1985 regulations, it is industry best practice).
- Formulation: A full qualitative list of ingredients.
Once submitted, the notification is added to the database. Note that this is not an approval or endorsement by Medsafe. It is a declaration by the sponsor that the product complies with the law. Medsafe audits this database, and non-compliant products can be recalled.
Considerations for Rongoā Māori
For the NZ Integrated Herbal Medicine & Rongoā Māori Hub, which also provides insights into Cupping Therapy: Inflammation and Recovery, understanding the intersection of regulation and traditional Māori healing (Rongoā) is vital. Rongoā Māori is protected under the Treaty of Waitangi (Te Tiriti o Waitangi).
Traditionally prepared Rongoā provided by a practitioner to a patient is often distinct from a mass-produced commercial product sitting on a retail shelf. However, when Rongoā products are commercialized, packaged, and sold to the general public (without a consultation), they generally fall under the Dietary Supplements Regulations or the Medicines Act depending on the claims made.
Practitioners should be careful with labelling. While traditional knowledge validates the efficacy of certain plants (like Kawakawa or Kumarahou) or research on NZ Native Berries: Antioxidant Properties & Health, commercial labels must avoid strict therapeutic claims unless the product is registered as a medicine. The industry is currently advocating for a regulatory model that better respects Indigenous knowledge systems without imposing Western pharmaceutical frameworks on traditional taonga (treasures).

Required Documentation and Labelling
Documentation is your primary defense during an audit. Even though you do not submit a dossier for approval, you must hold a “Site Master File” or equivalent records containing:
- Certificates of Analysis (CoA) for all raw materials.
- Stability testing data proving the product lasts as long as the expiry date says.
- Evidence supporting any health claims made on the label.
Labelling Requirements
Labels must be in English and clearly legible. According to the regulations, your label must include:
- The common name of the product.
- A statement of the net weight or volume (or number of capsules).
- A complete list of ingredients (active and inactive).
- The name and address of the NZ sponsor/distributor.
- Storage instructions.
- Batch number and expiry date.
- Mandatory Warning: If the product is not a food, it is wise to include “Dietary Supplement.”
Failure to label correctly is the most common reason for product recalls in the NZ natural health sector.
Fees and Processing Timelines
One of the advantages of the New Zealand market compared to Australia (TGA) or Canada (Health Canada) is the lower barrier to entry regarding costs.
Government Fees
Currently, there is no fee charged by Medsafe to submit a notification to the Electronic Notification of Dietary Supplements database. This is a significant benefit for small businesses and startups. However, this may change if new legislation is enacted, so businesses should monitor Ministry of Health announcements.
Processing Time
The electronic notification is almost immediate. Once you click submit, the product appears in the database shortly after. However, the preparation time is where the work lies. You should allocate 4-8 weeks for:
- Label compliance review (hiring a consultant is recommended).
- Ingredient verification.
- Establishing the local sponsor agreement.
Maintaining Compliance Post-Registration
Registering (notifying) your natural health product is not the end of the road; it is the beginning of your liability. The Ministry of Health conducts market monitoring.
Adverse Reaction Reporting
As a sponsor, you are responsible for monitoring the safety of your product. If a consumer reports a severe adverse reaction, you must investigate and, if necessary, report this to the Centre for Adverse Reactions Monitoring (CARM). Ignoring safety signals can lead to prosecution under the Food Act or Medicines Act.
Advertising Standards
Your marketing materials (website, social media, brochures) are considered an extension of your label. You cannot make therapeutic claims on Instagram that you are forbidden from making on the bottle. The Advertising Standards Authority (ASA) actively polices this space. Ensure your marketing team understands the difference between “supporting health” and “treating disease.”

People Also Ask
Do I need a license to sell herbal tea in NZ?
If the herbal tea is sold purely as a food (e.g., Peppermint tea for enjoyment), it falls under general Food Standards. If you market it with health claims (e.g., “Detox Tea” or “Sleep Aid”), it may fall under Dietary Supplements regulations, requiring notification and compliance with labelling rules.
Can I sell my NZ registered supplement in Australia?
Not automatically. Australia has a much stricter system governed by the TGA (Therapeutic Goods Administration). You generally need to list your product on the ARTG (Australian Register of Therapeutic Goods), which requires a separate application, fees, and GMP evidence.
How much does it cost to register a supplement in NZ?
There is currently no government fee for the electronic notification of a dietary supplement. However, businesses should budget for consultancy fees, label design, and testing (stability/microbial), which can range from $2,000 to $10,000+ depending on complexity.
What happens if I make therapeutic claims on a supplement?
If you make therapeutic claims without consent, Medsafe may classify your product as an unapproved medicine. This can lead to product seizure, recalls, fines, and prosecution under the Medicines Act 1981.
Is CBD oil a dietary supplement in NZ?
No. In New Zealand, CBD products are generally classified as prescription medicines or controlled drugs depending on their composition. They cannot be sold as over-the-counter dietary supplements under the current regulations.
Do I need GMP certification for NZ supplements?
While the Dietary Supplements Regulations 1985 do not explicitly mandate GMP (Good Manufacturing Practice) in the same way the Medicines Act does, operating without it is a massive risk. GMP is the industry standard for ensuring safety and quality, and it is often required by retailers and for export.