Herbal product labeling NZ refers to the strict packaging and information standards enforced by Medsafe and the Ministry for Primary Industries (MPI) under the Dietary Supplements Regulations 1985 and the Food Act 2014. To ensure compliance, manufacturers must clearly display the product name, a complete quantitative list of active ingredients, accurate dosage instructions, batch numbers, expiry dates, and mandatory allergen warnings, while avoiding unapproved therapeutic claims. For more information, visit our Home page.
The Regulatory Landscape for NZ Herbal Products
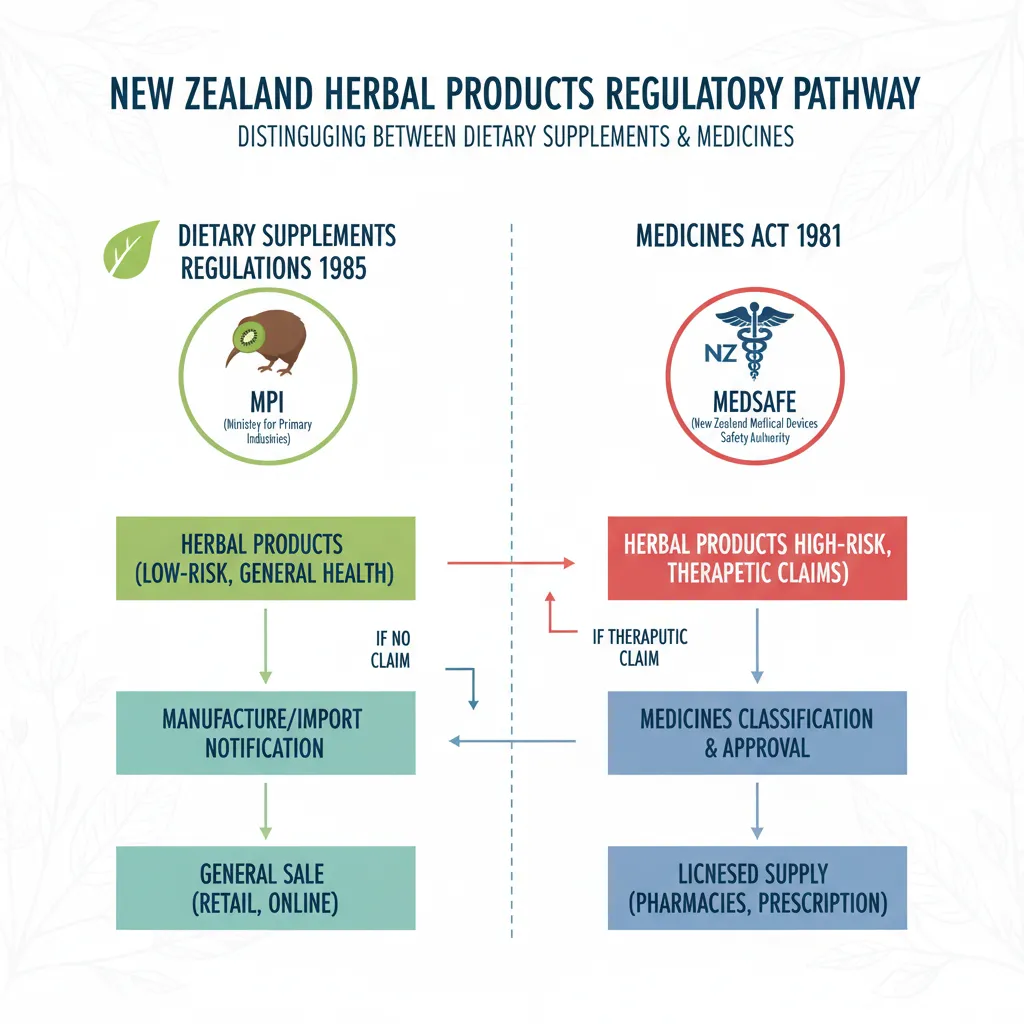
Navigating the regulatory environment for herbal medicine in New Zealand requires a deep understanding of the intersection between food safety laws and medicinal regulations. Unlike some jurisdictions where herbal products have a standalone category, New Zealand currently regulates most herbal products and natural health supplements under the Dietary Supplements Regulations 1985, which falls under the Food Act 2014, or as medicines under the Medicines Act 1981 depending on the claims made.
For the vast majority of herbal product manufacturers, the goal is to market the product as a dietary supplement. This classification allows for easier market entry but comes with strict limitations on what can be said about the product. The primary regulator involved is Medsafe New Zealand, which oversees the classification of products to ensure they do not cross the line into becoming unapproved medicines. Simultaneously, the Ministry for Primary Industries (MPI) ensures that these products meet food safety standards.
It is critical for manufacturers to understand that if a label makes a therapeutic claim—stating that the herb treats, cures, or prevents a disease—the product is legally classified as a medicine. This triggers a requirement for a rigorous consent process. Therefore, the art of herbal product labeling in NZ is ensuring the consumer understands the benefits of the product without making prohibited medical claims.
Mandatory Information: What Must Be on the Label
Under the Dietary Supplements Regulations 1985, specific information is non-negotiable. Omitting any of these elements can result in product recalls, fines, and significant reputational damage. Below is the comprehensive checklist for compliant herbal product labeling.
1. The Common Name of the Product
The label must clearly state the common name of the product. This should be descriptive enough that a consumer understands what the product is (e.g., “Kawakawa Balm” or “Echinacea Throat Spray”).
2. Statement of Ingredients
Transparency is paramount. You must list the active ingredients and their quantities. For herbal products, this typically involves stating the botanical name (Latin binomial) and the part of the plant used (e.g., root, leaf, flower).
- Quantitative declaration: You must state the amount of the active ingredient per dose (e.g., “Each 5ml contains 200mg of dry root equivalent”).
- Excipients: Non-active ingredients (fillers, binders, preservatives) must also be listed, usually in descending order of weight.
3. Dosage and Usage Instructions
Clear instructions on how to use the product, such as for Ignatia Amara: Grief and Emotional Stress, are mandatory. This includes the recommended daily dose, the method of administration (e.g., “Take one capsule daily with food”), and the target demographic (e.g., “Adults”). If the product is not suitable for children, this must be explicitly stated.
4. Batch Number and Expiry Date
Traceability is a cornerstone of consumer safety. Every unit sold must carry a unique batch number to facilitate recalls if necessary. Furthermore, an expiry date or “Best Before” date must be printed to indicate the shelf life during which the product remains effective and safe.
5. Name and Address of the Sponsor
The label must include the physical address of the manufacturer, packer, or the New Zealand distributor (sponsor). A website or PO Box alone is generally insufficient for the primary legal address requirement; a physical location ensures accountability.
Warning Statements and Allergen Declarations
Consumer safety extends beyond knowing what is in the bottle; it is equally about knowing who should not take the product. New Zealand adheres to strict standards regarding allergen declaration, aligning closely with Food Standards Australia New Zealand (FSANZ) requirements.
Mandatory Warning Statements
Certain herbs and supplements require specific warnings. For example, products containing St. John’s Wort must carry warnings regarding interactions with prescription medicines. High-dose Vitamin A products require pregnancy warnings. A general safety statement such as “Always read the label and use as directed. If symptoms persist, see your healthcare professional” is standard industry best practice and often required depending on the classification.
Allergen Declarations
If your herbal product is manufactured in a facility that handles common allergens, or if the product contains them, they must be declared. The “Big 8” allergens (including gluten, crustaceans, eggs, fish, peanuts, soybeans, milk, and tree nuts) must be emphasized. For herbal tinctures that use an ethanol base derived from wheat, or capsules derived from gelatin (animal product), clear labeling is essential for consumer choice and safety.
Labeling Considerations for Rongoā Māori
For producers of Rongoā Māori (traditional Māori medicine), labeling presents a unique opportunity to honor cultural heritage while meeting Western regulatory frameworks. The resurgence of Rongoā has led to a market needing clear guidance on how to present traditional knowledge respectfully, including principles of Ethical Sourcing and Sustainable Harvesting of Rongoā Plants.
When labeling Rongoā products, it is acceptable and encouraged to use Te Reo Māori names for plants (e.g., Kumarahou, Manuka). However, to ensure compliance with the Dietary Supplements Regulations, the scientific (Latin) name should also be provided in the ingredients list. This “dual-naming” approach satisfies regulatory inspectors while maintaining cultural integrity.
Furthermore, traditional use claims must be carefully phrased. Instead of claiming a product “cures eczema,” a compliant label might state the product contains herbs “traditionally used in Rongoā Māori to soothe the skin.” This distinguishes the product as utilizing traditional knowledge rather than making a verified pharmaceutical claim.

Navigating Therapeutic Claims and Advertising
The most common pitfall for NZ herbal businesses is the use of therapeutic claims. Under the Medicines Act 1981, a product is deemed a medicine if it claims to treat, cure, prevent, or diagnose a disease. If you have not registered your product as a medicine (a costly and lengthy process), you cannot make these claims.
Permitted vs. Prohibited Claims
- Prohibited (Medical): “Cures insomnia,” “Treats arthritis,” “Prevents flu,” “Anti-inflammatory.”
- Permitted (Functional/Supportive): “Supports restful sleep,” “Supports joint mobility,” “Supports immune system health,” “Soothes dry throats.”
These rules apply to the physical label, the packaging insert, your website, and your social media. Regulators view all these mediums as part of the product “advertisement.” A compliant label on the bottle does not protect you if your website claims the product cures cancer.
Balancing Brand Identity with Regulatory Compliance
Packaging is often the first touchpoint a customer has with your brand. The challenge lies in integrating the dense regulatory text without cluttering the design or diluting the brand aesthetic. High-quality herbal product labeling NZ requires a synergy between the regulatory affairs team and the graphic design team.
Legibility and Font Size
Regulations often dictate that mandatory information must be “conspicuous.” While specific font sizes can vary based on the container size, a general rule of thumb is that mandatory text should be no smaller than 1.5mm in height for the lowercase ‘o’. Key information like the product name and net weight must be prominent. Using high-contrast colors (e.g., black text on a white background) for the ingredients list is highly recommended to ensure accessibility for visually impaired consumers.
Many successful brands utilize “peel-and-reveal” labels or outer cartons to provide ample space for storytelling and branding while reserving the immediate bottle label for critical compliance data. This approach is particularly useful for small bottles (10ml-30ml) often used for essential oils or tinctures.

Best Practices for Clear and Informative Labeling
Beyond the legal minimums, adhering to best practices builds trust and authority with your customer base. In the Integrated Herbal Medicine & Rongoā Māori Hub, trust is the currency of success.
1. Sustainability Credentials
NZ consumers are environmentally conscious. If your packaging is recyclable, compostable, or made from recycled materials, label it clearly. However, ensure these claims are substantiated to avoid “greenwashing.”
2. Origin Labeling
While “Made in New Zealand” is a powerful marketing tool, it is also regulated. Ensure you meet the criteria for substantial transformation if you are importing ingredients but manufacturing the final dosage form in NZ. Using the official “New Zealand Made” FernMark (if licensed) adds significant credibility.
3. Standardization Declarations
For high-quality herbal products, stating the standardization of the extract (e.g., “Standardized to contain 5% withanolides”) signals potency and quality to educated consumers. This moves the product from a generic supplement to a professional-grade herbal medicine.
For further reading on the specific legislation governing these requirements, refer to the New Zealand Legislation website regarding the Dietary Supplements Regulations 1985.
People Also Ask
Do I need a license to sell herbal tea in NZ?
Generally, you do not need a specific pharmaceutical license to sell herbal tea if it is marketed as a food or dietary supplement and makes no therapeutic claims. However, you must register your business under the Food Act 2014 through the Ministry for Primary Industries (MPI) to ensure food safety compliance.
What are the labeling requirements for dietary supplements in NZ?
Dietary supplements must label the common name, a list of active ingredients and their quantities, the dose form, batch number, expiry date, name and address of the sponsor, and mandatory warning statements. They must not claim to treat or cure diseases.
Can I make therapeutic claims on herbal products?
No, not unless the product is registered as a medicine with Medsafe. You are limited to “structure/function” claims, such as “supports immune health,” rather than medical claims like “cures the flu.”
How do I declare allergens on herbal labels?
You must declare any common allergens (peanuts, tree nuts, milk, eggs, soy, wheat, fish, crustaceans) present in the product. These should be clearly listed in the ingredients panel or a separate “Contains” statement to warn consumers.
What is the difference between a medicine and a dietary supplement in NZ?
The main difference lies in the claims and the intent. Medicines are for treating or preventing disease and require Medsafe consent. Dietary supplements are for maintaining health and supplementing the diet, regulated under the Food Act and Dietary Supplements Regulations.
Does Rongoā Māori follow the same labeling rules?
Yes, if Rongoā Māori products are sold commercially as dietary supplements, they must adhere to the Dietary Supplements Regulations 1985 regarding labeling, ingredients lists, and safety warnings, regardless of their traditional origins.



