Medsafe herbal claims regulations dictate that unless a product is registered as a medicine, it cannot advertise a therapeutic purpose, such as treating, preventing, or curing a disease. Manufacturers must distinguish between “dietary supplements” (supporting normal health) and “medicines” (altering physiological function) to comply with the Medicines Act 1981 and avoid enforcement action.
The Regulatory Framework: Medicines vs. Supplements
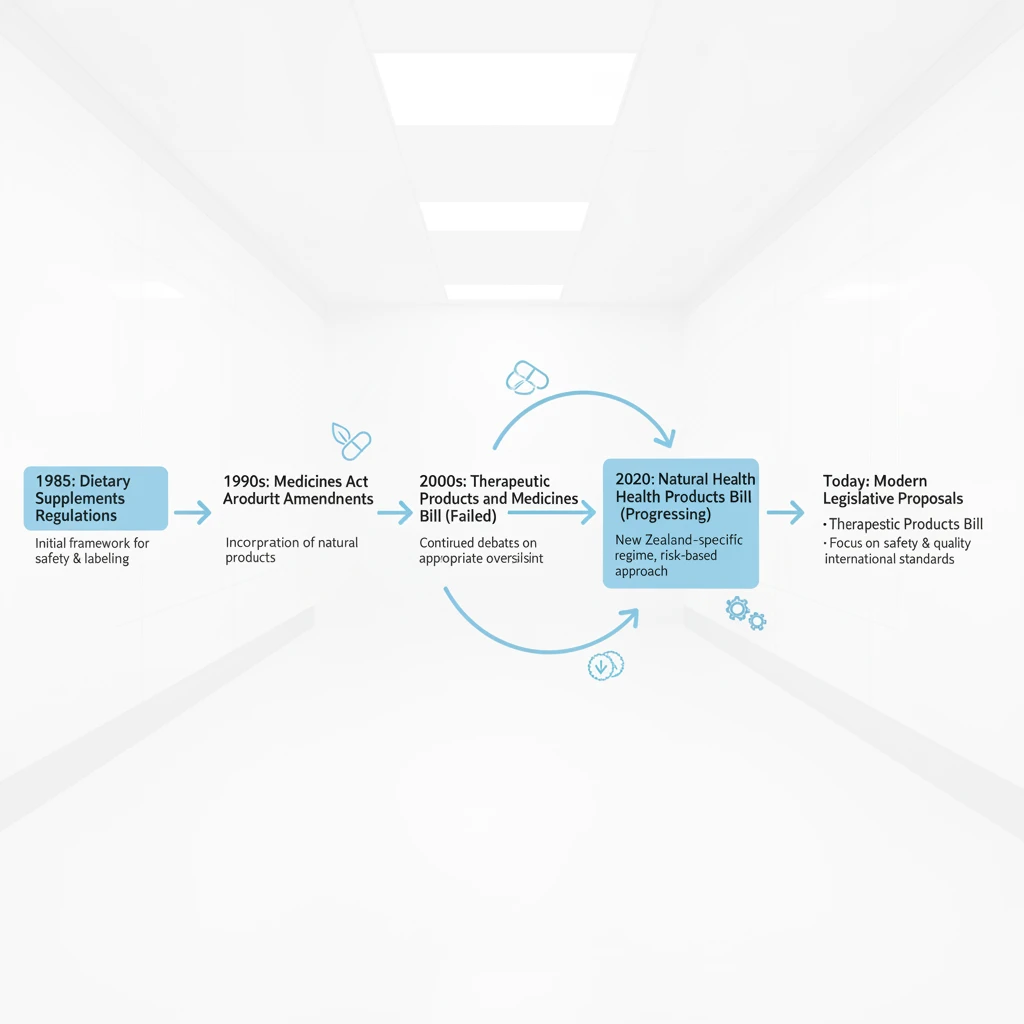
In New Zealand, the landscape for herbal medicine and natural health products is governed primarily by the Medicines Act 1981 and the Dietary Supplements Regulations 1985. For stakeholders in the Integrated Herbal Medicine & Rongoā Māori Hub, understanding where a product falls within this legislative spectrum is the first step toward compliance.
Medsafe, the New Zealand Medicines and Medical Devices Safety Authority, is responsible for administering the Medicines Act. Their primary concern is public safety. Return to our Home for more info. If a product makes a claim to treat, diagnose, prevent, or cure a disease, it is legally classified as a medicine. Consequently, it must undergo a rigorous assessment and registration process to prove its safety, quality, and efficacy before it can be sold.
Conversely, most herbal products on the New Zealand market are sold as “dietary supplements.” Under the Dietary Supplements Regulations 1985, a supplement is defined as a product intended to supplement the intake of food. These products are not permitted to claim a therapeutic purpose. The tension between wanting to explain the benefits of a powerful herb and the legal restriction against making therapeutic claims is the central challenge for marketers in this niche.

Defining “Therapeutic Purpose”
The definition of “therapeutic purpose” is broad under Section 4 of the Medicines Act. It includes:
- Treating or preventing disease.
- Diagnosing disease or ascertaining the existence of a physiological condition.
- Altering the shape, structure, size, or weight of the human body.
- Otherwise preventing or interfering with the normal operation of a physiological function.
If your advertising material, packaging, or website content implies any of the above, Medsafe may deem your herbal product an unapproved medicine. This can lead to product recalls, fines, and prosecution.
Permissible vs. Impermissible Health Claims
Navigating Medsafe herbal claims requires a nuanced understanding of language. The difference between a compliant claim and a non-compliant one often comes down to specific verbs and the context in which they are used.
Impermissible Therapeutic Claims
You cannot make claims that link your product to a specific disease or a state of disease. Even if there is clinical evidence supporting the herb’s efficacy, you cannot state it on the label unless the product is a registered medicine. Examples of high-risk words and phrases include:
- “Cures insomnia”
- “Treats arthritis”
- “Prevents colds and flu”
- “Anti-inflammatory” (implies treating inflammation, a medical condition)
- “Relieves pain”
Permissible Structure-Function Claims
For dietary supplements, you are generally restricted to “structure-function” claims. These describe how a nutrient or herb supports the normal maintenance of the body. These claims must assume the user is healthy and that the product helps maintain that state of health.
Acceptable phrasing often uses words like “supports,” “maintains,” “aids,” or “helps.” Examples include:
- Instead of “Cures Insomnia,” use “Supports natural sleep patterns” or “Supports relaxation.”
- Instead of “Treats Arthritis,” use “Supports joint mobility” or “Maintains joint health.”
- Instead of “Prevents Flu,” use “Supports immune system health.”
It is critical to note that simply adding the word “supports” does not automatically make a claim compliant if the rest of the sentence describes a disease state. For example, “Supports the body during cancer treatment” is still a therapeutic claim because it references cancer.
Evidential Requirements for Product Benefits
While dietary supplements do not require pre-market approval from Medsafe in the same way medicines do, this does not mean you can make baseless claims. Under the Fair Trading Act 1986, all representations made in trade must be truthful, accurate, and capable of being substantiated.
Substantiation of Claims
If you claim that your herbal product “Supports digestive health,” you must hold evidence to back that up. This evidence usually takes the form of:
- Traditional use evidence (documented history of use in herbal medicine text).
- Scientific literature (peer-reviewed studies on the specific ingredients).
- Clinical trials (though rare for supplements due to cost).
For more detailed information on the legislative requirements, you can refer to the Medsafe Authority website.
The Commerce Commission enforces the Fair Trading Act. If they question your claims, you must produce the evidence you held at the time the claim was made. You cannot go looking for evidence after an investigation begins.

Navigating Rongoā Māori and Traditional Medicine
For the NZ Integrated Herbal Medicine & Rongoā Māori Hub, the intersection of traditional Māori healing and modern regulation is particularly significant. This includes traditional practices like Rongoā for Emotional and Spiritual Wellbeing, as Rongoā Māori is a holistic system that is recognized as a taonga (treasure) under the Treaty of Waitangi.
The Practitioner Exemption
Section 28 of the Medicines Act 1981 provides an exemption for herbalists and naturopaths. It allows practitioners to manufacture and supply medicines to individuals following a consultation, which can involve techniques like Tincture Making with New Zealand Herbs: Alcohol & Glycerine Extracts. This means a Rongoā practitioner can prepare a specific remedy for a patient’s ailment and discuss its therapeutic purpose during the consultation.
However, this exemption does not apply to mass-marketed products. Once a Rongoā product is bottled, labeled, and placed on a retail shelf (or sold online to the general public without a consultation), it falls under the general regulations. It must be sold as a dietary supplement (without therapeutic claims) or registered as a medicine.
Marketers of Rongoā products must be careful not to exploit traditional knowledge in a way that violates Medsafe’s guidelines. For instance, citing traditional use to justify a claim like “Historically used to treat infection” on a retail label is considered a therapeutic claim and is non-compliant.
Advertising Standards and Consumer Protection
Beyond Medsafe and the Commerce Commission, the Advertising Standards Authority (ASA) maintains the Therapeutic and Health Advertising Code. This code applies to all advertisements for health products and services.
Key ASA Principles
The ASA Code requires a high standard of social responsibility. Key rules include:
- Truthful Presentation: Advertisements must not be misleading or deceptive.
- Scientific Terminology: Jargon should not be used to confuse consumers or imply a scientific validity that does not exist.
- Testimonials: Testimonials must be genuine, but they cannot be used to make claims that the advertiser cannot make themselves. If a customer writes, “This cured my eczema,” and you publish that review on your website, you have made a non-compliant therapeutic claim.
For further reading on consumer protection laws, refer to the Fair Trading Act 1986 legislation.
Best Practices for Avoiding Misleading Marketing
To operate a successful and legal herbal business in New Zealand, strict adherence to compliance protocols is necessary. Below is a strategy to ensure your marketing materials remain within the law while still effectively communicating value.
1. The “Is it a Disease?” Test
Review every sentence on your packaging and website. Ask yourself: “Does this word describe a disease, an ailment, or a defect of the body?” If the answer is yes, remove it. Focus on health maintenance.
2. Context Matters
Medsafe looks at the overall presentation. The name of the product, the imagery used (e.g., a picture of a heart), and the text all contribute to the “intended purpose.” A tea named “Flu-Buster” implies a therapeutic purpose by name alone, even if the description only says “supports immunity.”
3. Disclaimer Usage
While disclaimers like “This product is not intended to diagnose, treat, cure, or prevent any disease” are standard (and often required for exports to the USA), they do not immunize you against NZ law. If you make a therapeutic claim in the headline and try to disclaim it in the footer, you are still liable for a breach of the Medicines Act.
4. Regular Audits
Regulations and enforcement priorities can change. Conduct an annual audit of your digital footprint, including social media posts. Old posts that make non-compliant claims can still trigger enforcement action.

Conclusion
Navigating Medsafe herbal claims is a complex but essential task for any business in the NZ Integrated Herbal Medicine & Rongoā Māori Hub. By understanding the distinction between medicines and dietary supplements, respecting the boundaries of the Medicines Act 1981, and adhering to ethical advertising standards, you can build a trustworthy brand that serves the community safely. Always prioritize clear, structure-function claims backed by evidence, and when in doubt, seek professional regulatory advice.
People Also Ask
What claims can I legally make about herbal products in NZ?
In New Zealand, unless your product is a registered medicine, you can only make “structure-function” claims. These describe how the herb supports or maintains normal health and physiological function (e.g., “Supports joint health”). You cannot claim to treat, cure, or prevent any disease.
Does Medsafe regulate Rongoā Māori products?
Medsafe regulates Rongoā Māori products if they are sold to the public with therapeutic claims. However, under Section 28 of the Medicines Act, practitioners are generally exempt when preparing remedies for individual patients following a consultation.
What is the difference between a dietary supplement and a medicine?
A medicine is a substance with a therapeutic purpose (treating/preventing disease) and requires registration. A dietary supplement is intended to supplement the diet and support normal health but cannot claim to alter the body’s physiology or treat illnesses.
Can I use customer testimonials that mention curing a disease?
No. Using a customer testimonial that claims a product cured or treated a condition is considered a therapeutic claim made by the advertiser. This violates the Medicines Act and advertising codes.
What happens if I make non-compliant herbal claims?
Medsafe can order you to stop selling the product, recall it, or correct the advertising. Serious breaches can result in prosecution under the Medicines Act 1981, leading to significant fines or imprisonment.
Do I need to register my herbal product with Medsafe?
If you intend to market your product as a dietary supplement without therapeutic claims, you do not need to register it with Medsafe. However, if you want to make therapeutic claims, you must register it as a medicine.