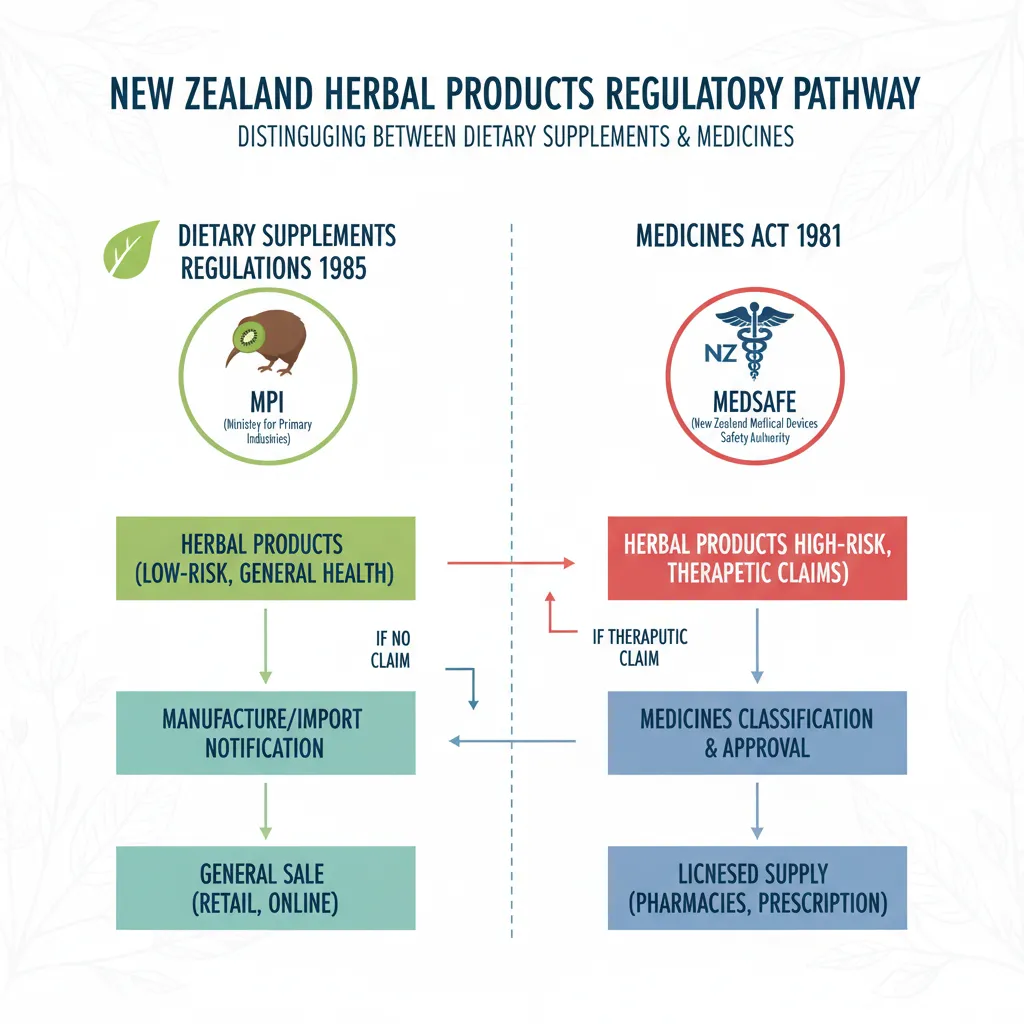
Medicine vs natural health product NZ is a distinction defined primarily by therapeutic claims and regulatory oversight at Home. Under the Medicines Act 1981, medicines are substances assessed and approved by Medsafe to treat, cure, or prevent disease using clinical evidence. Conversely, natural health products (NHPs), often regulated under the Dietary Supplements Regulations 1985, are intended to support general health and nutrition and cannot legally claim to treat medical conditions without specific consent.
Legal Definitions and Classifications in New Zealand
Navigating the healthcare landscape in Aotearoa New Zealand requires a clear understanding of the legislative boundaries that separate pharmaceutical interventions from holistic wellness supports. For consumers, practitioners, and manufacturers alike, the distinction between a “medicine” and a “natural health product” (NHP) is not merely semantic—it dictates availability, safety monitoring, and legal liability.
Defining a Medicine
Under the Medicines Act 1981, a medicine is broadly defined as any substance or article that is manufactured, imported, sold, or supplied wholly or principally for a therapeutic purpose. A “therapeutic purpose” includes treating or preventing disease, diagnosing conditions, or altering the physiological function of the body.
Medicines in New Zealand are categorized into three main tiers based on risk:
- Prescription Medicines: High-risk substances requiring a doctor’s authorization (e.g., antibiotics, antihypertensives).
- Pharmacist-Only Medicines (Restricted): Available without a prescription but only after consultation with a pharmacist (e.g., sildenafil, some antihistamines).
- Pharmacy-Only and General Sale Medicines: Lower-risk items available in pharmacies or supermarkets (e.g., paracetamol, small packs of ibuprofen).
Defining Natural Health Products (NHPs)
New Zealand currently lacks a specific, standalone “Natural Health Products Act” that is fully operational in the way the Medicines Act functions. Instead, most products that consumers identify as NHPs—such as vitamins, minerals, herbal extracts, and omega-3 oils—are regulated under the Dietary Supplements Regulations 1985 (under the Food Act).
A dietary supplement is defined as an amino acid, edible substance, herb, mineral, synthetic nutrient, or vitamin intended to supplement the diet, including remedies for Holistic Management of Insomnia. Crucially, because they are technically regulated closer to foods than drugs, they are not subjected to the same pre-market efficacy evaluation as medicines, provided they do not cross the line into making therapeutic claims, such as those related to Herbal Support for Menopause.

Regulatory Pathways: Medsafe vs. Food Standards
The journey from a raw ingredient to a product on a shelf differs vastly depending on whether the item is classified as a medicine or a natural health product. This divergence is overseen by different regulatory bodies with distinct mandates.
The Medsafe Approval Process
For a product to be sold as a medicine in New Zealand, it must undergo a rigorous evaluation by Medsafe (the New Zealand Medicines and Medical Devices Safety Authority). This process involves:
- Clinical Trials: Submission of extensive data proving the drug is effective for its intended indication.
- Safety Profiling: Detailed analysis of side effects, contraindications, and toxicity.
- Quality Assessment: Validation of the chemical purity and stability of the product.
This pre-market approval acts as a gatekeeper. If a product fails to demonstrate that its benefits outweigh its risks, it cannot enter the market. This process can take years and cost millions of dollars, ensuring a high barrier to entry that prioritizes public safety.
The Dietary Supplement Pathway
In contrast, natural health products regulated as dietary supplements do not require pre-market approval from Medsafe. There is no requirement to prove efficacy through clinical trials before sale. Instead, the responsibility lies heavily on the sponsor (the manufacturer or importer) to ensure the product is safe and complies with the list of permitted ingredients.
However, this does not mean the market is the “Wild West.” Products must still adhere to the Food Standards Code regarding composition and labeling. If a dietary supplement is found to contain undeclared prescription ingredients (a common issue with some imported weight-loss or libido products), Medsafe will intervene, classify it as an unapproved medicine, and order its removal.
Manufacturing Standards and Quality Assurance
One of the most critical, yet often invisible, differences between medicines and NHPs is the standard of manufacturing required by law.
Good Manufacturing Practice (GMP)
Medicines: It is mandatory for all medicines sold in New Zealand to be manufactured in facilities that are audited and certified for Good Manufacturing Practice (GMP). GMP ensures that products are consistently produced and controlled according to quality standards. It covers every aspect of production, from the starting materials and premises to the training and personal hygiene of staff.
Natural Health Products: For dietary supplements sold domestically, GMP certification is generally voluntary, though highly recommended. Many reputable NZ manufacturers voluntarily adhere to GMP standards (often to facilitate export to markets like Australia, where regulations are stricter). However, because it is not strictly mandatory for all domestic dietary supplements, there can be variability in product quality, potency, and purity across the market.

Marketing Limitations and Permissible Claims
The “therapeutic claim” is the legal tripwire that separates a dietary supplement from an unapproved medicine. This distinction causes the most confusion for consumers and frustration for herbalists.
The Forbidden Words
Under the Medicines Act, only an approved medicine can claim to:
- Treat
- Cure
- Prevent
- Diagnose
- Alleviate a symptom of a disease
If a bottle of Vitamin C claims to “cure the common cold,” it is breaking the law unless it has been registered as a medicine. Because the cost of registration is prohibitive for generic natural substances, manufacturers must use “soft” claims.
Permissible Structure-Function Claims
Natural health products are restricted to claims that describe how the nutrient or herb supports the normal structure or function of the body. They focus on maintenance and wellbeing rather than disease intervention.
- Medicine Claim: “Relieves arthritis pain and reduces inflammation.”
- NHP Claim: “Supports joint mobility and comfort.”
- Medicine Claim: “Treats insomnia and anxiety.”
- NHP Claim: “Supports relaxation and healthy sleep patterns.”
This linguistic dance ensures that consumers are not misled into thinking a supplement has the same proven clinical potency as a pharmaceutical, but it can also make it difficult for genuine herbal products to communicate their traditional uses.
Rongoā Māori in the Regulatory Landscape
In the context of Aotearoa, any discussion on health regulation must address Rongoā Māori (traditional Māori healing). Rongoā is holistic, encompassing not just rākau (herbal remedies) but also mirimiri (massage), karakia (prayer), and spiritual connection.
The imposition of Western regulatory frameworks like the Medicines Act creates tension with Rongoā. Traditional practitioners generally do not manufacture products for mass market retail; they provide customary care to their whānau and hapū. However, when Rongoā products (like Kawakawa balm or Kumarahou tonic) are commercialized and sold in retail settings, they technically fall under the same Dietary Supplements or Cosmetics regulations as other NHPs.
There is a strong movement within New Zealand to ensure that any legislative changes protect the rights of Māori to practice Rongoā under Te Tiriti o Waitangi. This involves ensuring that the manufacture and supply of traditional remedies are not stifled by compliance costs designed for pharmaceutical giants, while still ensuring safety for the end user.
Consumer Perception and Safety Considerations
A prevailing myth in the consumer market is the “Naturalistic Fallacy”—the belief that because something is natural, it is inherently safe. This is incorrect. Natural health products contain bioactive compounds that can have potent physiological effects.
Drug-Herb Interactions
Because NHPs are often sold in supermarkets or health food stores without professional advice, the risk of interaction with prescribed medicines is significant. For example:
- St John’s Wort: Can significantly reduce the effectiveness of oral contraceptives, blood thinners, and antidepressants.
- Ginkgo Biloba: Can increase bleeding risk if taken with anticoagulants like warfarin.
- Grapefruit Seed Extract: Can alter the metabolism of statins and heart medications.
For the “Medicine vs Natural Health Product” debate, the safety net differs. Pharmacists are trained to check for interactions with medicines. Retail assistants in health food stores may not have the same level of clinical training, placing a higher burden of responsibility on the consumer to disclose their supplement use to their GP.

The Future of Health Regulation in NZ
The regulatory environment in New Zealand is currently in a state of flux. The Therapeutic Products Act 2023 was intended to replace the aging Medicines Act 1981 and Dietary Supplements Regulations 1985, creating a comprehensive scheme that would regulate medicines, medical devices, and natural health products under one umbrella.
However, with changes in government, the implementation of this Act has faced significant scrutiny, pauses, and potential repeal or amendment. The core tension remains: how to regulate low-risk natural products in a way that ensures quality and safety without imposing the crushing compliance costs associated with high-risk pharmaceuticals.
For the time being, the distinction remains anchored in the 1981 and 1985 legislations. Medicines treat disease and require proof; NHPs support health and require safety. For the Integrated Herbal Medicine & Rongoā Māori Hub, staying abreast of these changes is vital for compliance and for advocating for a system that respects both scientific rigour and traditional wisdom.
People Also Ask
Are natural health products regulated in NZ?
Yes, they are currently regulated primarily under the Dietary Supplements Regulations 1985 (under the Food Act) and the Medicines Act 1981. While they do not require the same pre-market clinical trials as medicines, they must be safe for consumption, contain only permitted ingredients, and cannot make therapeutic claims without consent.
Can I sell herbal medicine in NZ?
You can sell herbal products as dietary supplements provided you do not make therapeutic claims (e.g., “cures eczema”). If you make therapeutic claims, the product is classified as a medicine and requires Medsafe approval. Practitioners can also dispense herbs to patients following a consultation under specific exemptions.
What is the difference between a dietary supplement and a medicine?
The main difference is the intent and the claim. Medicines are for treating or preventing disease and require Medsafe approval based on clinical evidence. Dietary supplements are for maintaining general health and nutrition and are regulated closer to food standards.
Is Rongoā Māori considered a medicine?
Legally, if Rongoā products are sold with therapeutic claims, they fall under the Medicines Act. However, Rongoā is a taonga protected by the Treaty of Waitangi. Traditional practice is generally distinct from commercial manufacturing, but commercialized Rongoā products must comply with current consumer safety laws.
How do I know if a medicine is approved in NZ?
You can search for the product on the Medsafe website. Approved medicines will have a classification (e.g., Pharmacy Only, Prescription Only) and data sheets available. If a product is not listed there, it is likely unregulated or sold as a dietary supplement.
Can natural products interact with prescription drugs?
Yes, many natural products interact with prescription medicines. For example, St John’s Wort can interfere with birth control and antidepressants. It is crucial to consult a healthcare professional or pharmacist before combining natural health products with prescribed medication.