Herbal product consumer rights NZ are primarily protected under the Consumer Guarantees Act (CGA) and the Fair Trading Act. These laws ensure that natural health products and dietary supplements sold in New Zealand must be of acceptable quality, safe for consumption, and fit for their intended purpose. While many herbal remedies are regulated as dietary supplements rather than medicines, consumers legally deserve accurate labeling, truthful advertising, and products that match their description. For more information, visit our Home page.
The Regulatory Landscape: Supplements vs. Medicines
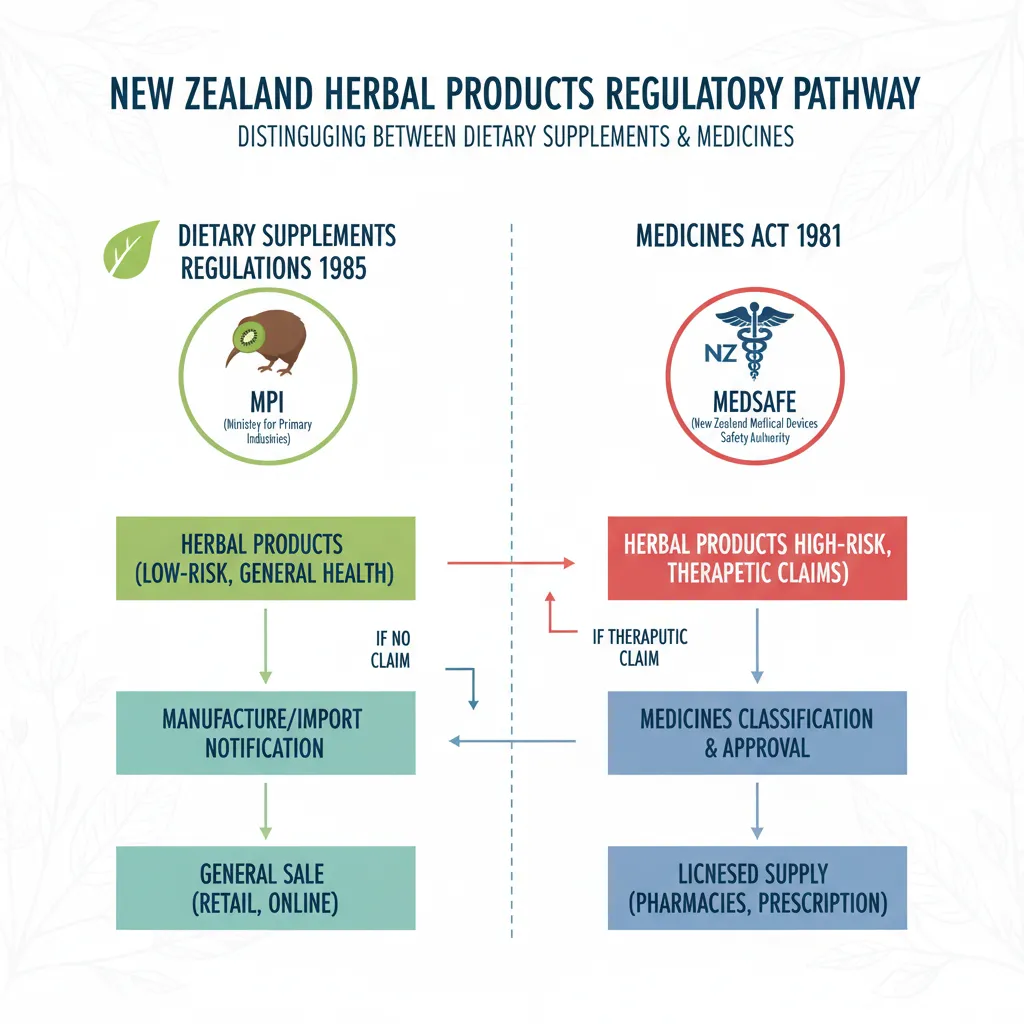
New Zealand has a unique regulatory environment for herbal products. Unlike pharmaceutical drugs, which undergo rigorous pre-market testing and approval by Medsafe, the majority of herbal products available on store shelves are classified as “dietary supplements” under the Dietary Supplements Regulations 1985. This distinction is critical for consumers to understand because it dictates the level of scrutiny a product receives before it reaches your hands.
Under current New Zealand law, a dietary supplement is defined as a product intended to supplement the diet, containing vitamins, minerals, or herbs. Suppliers are not permitted to make therapeutic claims—such as stating a product will “cure” or “treat” a specific disease—unless the product has been registered as a medicine. However, the line is often blurred in marketing materials, leading to consumer confusion.
Because these products are not subject to the same efficacy trials as pharmaceuticals, the onus often falls on the manufacturer to ensure safety and quality. This regulatory gap highlights the importance of being an informed consumer. While the government monitors safety post-market, knowing your rights and how to identify quality products is your first line of defense.
Decoding Product Labels and Ingredient Lists
One of the most powerful tools you have as a consumer is the ability to read and interpret a product label. In the herbal medicine sector, labeling can vary significantly between brands, but specific details serve as markers of quality and transparency.

Botanical Names vs. Common Names
High-quality herbal products will always list the botanical (Latin) name of the plant alongside the common name. For example, a label should read “Echinacea purpurea (Echinacea)” rather than just “Purple Coneflower.” This is vital because different species of the same plant genus can have vastly different chemical constituents and therapeutic effects, as demonstrated in Ashwagandha: Clinical Evidence for Stress & Cortisol. If a label lacks the botanical name, it is a red flag regarding the manufacturer’s professional standards.
Standardised Extracts vs. Whole Herb
You will often see terms like “Standardised Extract” or ratios like “10:1” on labels. A 10:1 extract means that 10 kilograms of raw herb were used to produce 1 kilogram of extract. Standardisation ensures that the capsule contains a guaranteed level of the active compound (marker compound). For consumers seeking consistency—ensuring the pill you take today works the same as the one you take next month—standardised extracts are generally preferred.
Excipients and Fillers
The ingredient list must also disclose non-active ingredients, known as excipients. These include binders, flow agents, and capsule shells. Consumers with allergies or dietary restrictions (such as vegans or those with gluten intolerance) must scrutinize this section. Common excipients include magnesium stearate and silicon dioxide. While generally safe, an excessively long list of fillers may indicate a lower-quality product.
Identifying Reputable Suppliers and Brands
Not all herbal products are created equal. In an industry where distinct regulations are still evolving, the reputation of the supplier is paramount. To ensure you are buying safe and effective products, look for affiliation with professional bodies.
The New Zealand Association of Medical Herbalists (NZAMH) sets high standards for practice and product quality. Manufacturers or practitioners aligned with such organizations are bound by codes of ethics and continuing education requirements. When purchasing from a naturopath or medical herbalist, ask about their registration and the source of their dispensing herbs.
Good Manufacturing Practice (GMP)
The gold standard for production quality is GMP certification. Manufacturers who adhere to GMP standards have rigorous protocols for hygiene, raw material testing, and record-keeping. Look for “GMP Certified” logos on product packaging. This certification provides assurance that what is on the label is actually in the bottle, free from contaminants like heavy metals or pesticides.
Your Rights Under the Consumer Guarantees Act
When you purchase herbal products in New Zealand, you are protected by the Consumer Guarantees Act 1993 (CGA). This Act applies to goods sold for personal use and provides a safety net if a product fails to meet expectations. It is important to note that the CGA applies to the retailer who sold you the product, not just the manufacturer.
Guarantee of Acceptable Quality
Under the CGA, herbal products must be of “acceptable quality.” This means they must be:
- Safe to use.
- Fit for all the purposes for which goods of that type are commonly supplied.
- Acceptable in appearance and finish.
- Free from minor defects.
If you buy a bottle of fish oil capsules and they are rancid (oxidized) upon opening, the product is not of acceptable quality. You have the right to return it to the retailer for a replacement or a refund.
Guarantee of Fitness for Particular Purpose
This is a critical aspect for herbal medicine consumers. If you ask a retailer or practitioner for a product to help with a specific issue (e.g., “I need something to help me sleep” or Herbal Support for Menopause), and they recommend a specific herb, they are guaranteeing that the product is fit for that purpose. If you take the product as directed and it has absolutely no effect or worsens the condition, you may have grounds for a remedy under the CGA, provided you relied on the seller’s expertise.
Misleading Claims and the Fair Trading Act
The Fair Trading Act prohibits misleading or deceptive conduct. If a product label claims it is “100% Organic” but contains non-organic fillers, or claims to be “Made in NZ” when it was only packaged here, the supplier is breaking the law. Consumers can report such breaches to the Commerce Commission.
Safety First: Adverse Reactions and Contraindications
“Natural” does not always mean “safe.” Herbal medicines contain potent chemical compounds that can interact with pharmaceutical drugs, affect surgery outcomes, or cause allergic reactions. Consumer safety rights include the right to be informed about these risks.

Herb-Drug Interactions
Some interactions are well-documented. For example, St John’s Wort can reduce the effectiveness of the contraceptive pill and blood thinners. Ginkgo biloba can increase bleeding risk. Consumers taking prescription medication should always consult a healthcare professional before starting a herbal regimen. If a product label fails to warn of known, serious interactions, this could be considered a safety defect.
Reporting Adverse Reactions
If you experience a negative reaction to a herbal product, you should stop taking it immediately. In New Zealand, you can report these reactions to the Centre for Adverse Reactions Monitoring (CARM). Reporting helps authorities track potential safety issues with specific batches or brands, protecting other consumers in the long run.
Cultural Safety and Rongoā Māori
In Aotearoa New Zealand, the consumer landscape includes Rongoā Māori (traditional Māori healing). When engaging with Rongoā, consumer rights intersect with cultural respect and intellectual property rights.
Authentic Rongoā is often holistic, involving wairua (spiritual) aspects alongside the physical application of plants like Kawakawa or Manuka. Commercialization of these taonga (treasures) has led to some mass-produced products that may lack the traditional tikanga (protocols) of harvesting and preparation. Consumers seeking authentic Rongoā should look for practitioners who are connected to their whakapapa and local iwi, ensuring that the knowledge and plants are treated with the appropriate respect. While the CGA still applies to purchased Rongoā products, the relationship is often viewed through a model of partnership and trust rather than a simple transaction.
Buying Online: Risks and Red Flags
The digital age has made global herbal markets accessible, but buying online significantly complicates your consumer rights. When you purchase from an overseas website, the New Zealand Consumer Guarantees Act does not apply. You are subject to the laws of the country where the seller is based, making it difficult to get a refund or replacement if things go wrong.

Customs and Biosecurity
New Zealand has strict biosecurity laws. Importing raw plant material, seeds, or honey products can lead to seizure at the border and significant fines. Furthermore, products manufactured overseas may contain ingredients that are illegal or prescription-only in New Zealand. Medsafe frequently issues alerts regarding international products found to contain hidden pharmaceutical ingredients (such as steroids or erectile dysfunction drugs) masquerading as “herbal supplements.”
To ensure your rights are protected and your safety is maintained, the best practice is to purchase from New Zealand-based retailers and manufacturers who are accountable under NZ law and adhere to local safety standards.
People Also Ask
Are herbal products regulated in New Zealand?
Yes, most herbal products are regulated as “dietary supplements” under the Dietary Supplements Regulations 1985. They must be safe and properly labeled, but they do not require the same pre-market approval as pharmaceutical medicines.
Can I return a herbal supplement if it doesn’t work?
Under the Consumer Guarantees Act, if a retailer specifically recommended a product for a certain purpose and it failed to achieve that purpose, you may be entitled to a remedy. However, if you chose the product yourself without advice, a lack of effectiveness is usually not grounds for a return unless the product is faulty.
How do I know if a herbal product is safe?
Look for products made by reputable NZ companies, ideally with GMP (Good Manufacturing Practice) certification. Check for clear labels listing botanical names and batch numbers, and consult the Medsafe website for any safety alerts.
Is Rongoā Māori covered by the Consumer Guarantees Act?
Yes, if Rongoā products or services are sold in trade (i.e., you pay for them), they are covered by the CGA. However, traditional exchanges or koha-based systems within a community context may function differently.
What should I do if I have an allergic reaction to a herbal product?
Stop taking the product immediately and seek medical advice. You should report the reaction to the Centre for Adverse Reactions Monitoring (CARM) and return the product to the retailer as it may be considered “unsafe” under the CGA.
Can I import herbal medicines into New Zealand?
You can import medicines for personal use, but strict rules apply. The product must be legal in NZ, not contain endangered species (CITES), and meet biosecurity standards. It is safer to buy from local suppliers to avoid seizure or fines.