The Therapeutic Products Act introduces a comprehensive regulatory framework for health products in New Zealand, profoundly impacting Rongoā Māori. While aiming to ensure product safety and efficacy, the legislation includes specific provisions and ongoing discussions to exempt traditional Rongoā practices, acknowledging their unique cultural and spiritual significance, while regulating commercial products derived from traditional knowledge.
Overview of the New Therapeutic Products Act
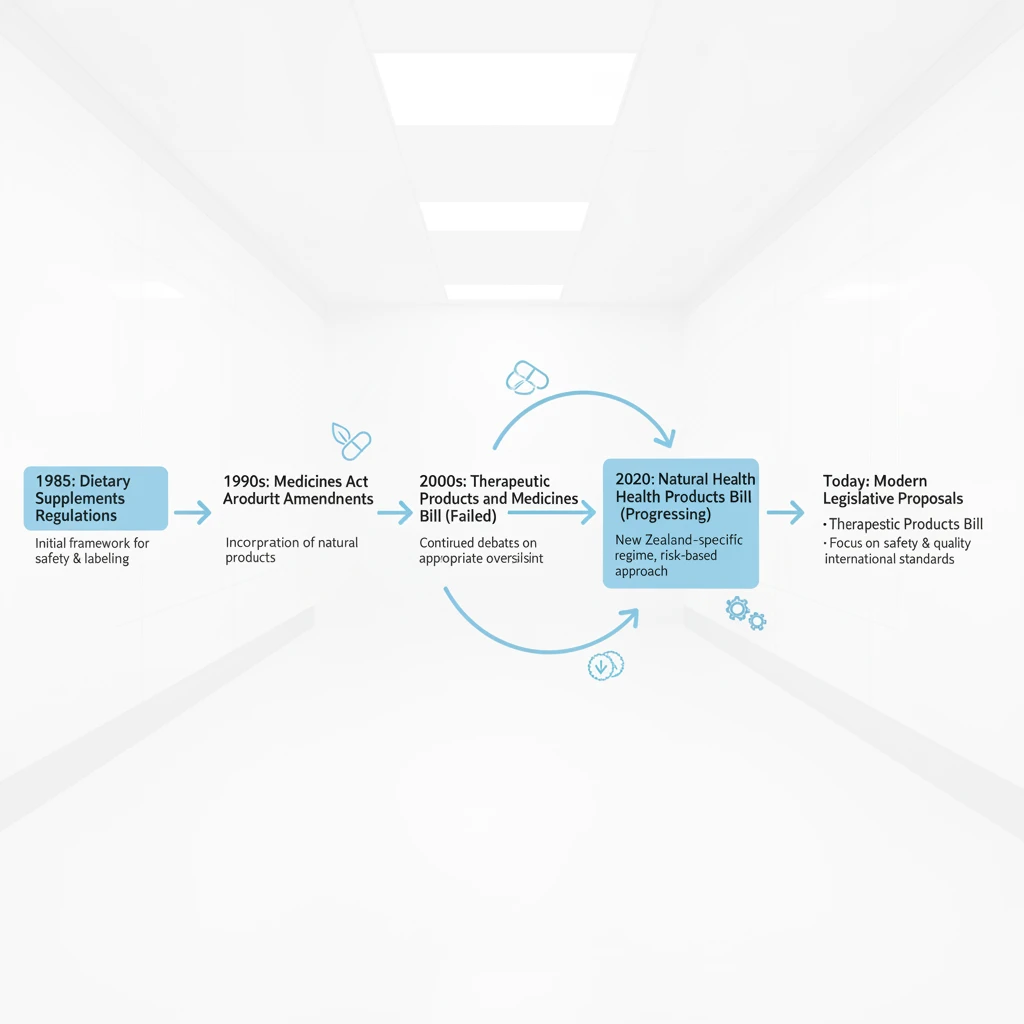
The Therapeutic Products Act, which received Royal Assent in July 2023, marks a monumental shift in how therapeutic products are regulated in Aotearoa New Zealand. Replacing the outdated Medicines Act 1981 and elements of other legislation, this new Act establishes a comprehensive, lifecycle-based regulatory system designed to ensure the safety, quality, and efficacy of a vast array of health products. These include medicines, medical devices, active pharmaceutical ingredients, and cell and tissue products, among others. The primary objectives are clear: protect public health, provide clarity for industry, and facilitate access to necessary treatments, while fostering innovation in the health sector. This legislation introduces a new regulator, the Therapeutic Products Branch, operating under the Ministry of Health, tasked with overseeing all aspects from product development and manufacturing to supply, advertising, and post-market surveillance. Its reach is extensive, touching virtually every product intended for a therapeutic purpose in the country. For the Rongoā Māori sector, understanding the nuances of this broad legislative sweep is paramount, as it presents both potential challenges and opportunities for the recognition and protection of traditional practices and products.
Historically, Rongoā Māori has operated largely outside conventional regulatory frameworks, guided instead by tikanga (Māori customs and protocols) and centuries of traditional knowledge. The previous regulatory landscape offered a degree of ambiguity, which, while allowing flexibility, also lacked explicit recognition and protection for traditional healing modalities. The new Act seeks to address this by explicitly considering traditional medicines and practices, aiming to strike a delicate balance between public safety and cultural preservation. It represents an opportunity to embed a bicultural approach into health regulation, recognising the unique status of Māori as Tangata Whenua and the inherent value of Rongoā Māori. The implementation of the Act will be phased, allowing time for various sectors to adapt to the new requirements, but the foundational principles are now firmly established. Stakeholders across the health landscape, from large pharmaceutical companies to small-scale Rongoā practitioners, must engage with this legislation to ensure continued compliance and culturally appropriate practice.
Central to the Act’s framework is a risk-based approach, where the level of regulation applied to a product is proportionate to the potential risks it poses to public health. This means that highly potent or invasive products will face stricter controls than those considered lower risk. This principle is particularly relevant for Rongoā Māori, where a diverse range of practices and products exist, from internal preparations and topical applications to spiritual healing and massage. The challenge, and indeed the focus of ongoing consultation, lies in determining how this risk-based approach can be applied in a culturally sensitive manner that respects the holistic nature of Rongoā. Furthermore, the Act includes provisions for therapeutic product standards, good manufacturing practices (GMP), and a robust compliance and enforcement regime. These elements are designed to instill confidence in the safety and quality of therapeutic products available to New Zealanders. For commercial entities producing Rongoā-inspired products, these standards will necessitate significant adjustments to their operational processes and quality management systems. The shift is not merely administrative; it reflects a fundamental re-evaluation of how health products are brought to market and sustained through their lifecycle in Aotearoa.

Exemptions and Protections for Traditional Practitioners
One of the most critical aspects of the Therapeutic Products Act, particularly for the Rongoā Māori community, concerns the provisions for exemptions and protections for traditional practices. The government has openly acknowledged the unique cultural status and significance of Rongoā Māori, recognising it as a taonga (treasure) under Te Tiriti o Waitangi. Consequently, the Act has been designed with an explicit intent to avoid over-regulating traditional Rongoā practices and practitioners. This commitment is articulated through specific clauses and through ongoing development of regulations that aim to create a clear distinction between the practice of Rongoā Māori as a traditional healing art and the commercial production of therapeutic products that may incorporate traditional elements.
Key to these protections is the understanding that many Rongoā practices involve holistic healing, which extends beyond the mere application of a physical product. This includes karakia (prayers), mirimiri (massage), and the use of waiata (songs), alongside the application of rongoā rākau (plant medicines). The Act’s focus is primarily on physical therapeutic products, and therefore, pure traditional Rongoā practices, when conducted by traditional practitioners within a customary context, are largely intended to be exempt from the extensive regulatory requirements for product approvals, manufacturing licenses, and advertising standards. The legislation explicitly allows for regulations to be made that exempt specific products or classes of products, and particular practices, from the scope of the Act, which is the mechanism intended to safeguard Rongoā Māori.
Furthermore, the Act facilitates the establishment of a Rongoā Māori advisory body. This body will play a pivotal role in informing the new regulator on matters pertaining to Rongoā Māori, ensuring that regulatory decisions are culturally informed and appropriate. This mechanism provides a crucial pathway for the voice of the Rongoā community to be heard and integrated into the regulatory framework, fostering a bicultural approach to health product governance. The involvement of traditional knowledge holders and practitioners in co-designing the specific exemptions and guidelines is vital to ensure that the protections are robust and genuinely reflective of Rongoā values and practices. This collaborative approach underscores the government’s commitment to upholding Te Tiriti o Waitangi principles within the health sector, ensuring that Māori self-determination (tino rangatiratanga) in health is respected and enabled.
Defining the Boundaries of Traditional Practice vs. Commercialisation
A significant challenge in implementing these exemptions lies in clearly defining the boundary between traditional Rongoā practice, which is intended to be exempt, and the commercial production of Rongoā-inspired products, which will likely fall under the Act’s regulatory scope. For instance, a traditional healer preparing a topical balm from plants gathered for a specific whānau (family) member’s ailment, used within a customary healing session, would generally be considered traditional practice. However, if that same balm is manufactured in larger quantities, packaged, marketed, and sold to the general public through retail channels, it would likely be viewed as a commercial therapeutic product and subject to the Act’s requirements. The distinction often hinges on the scale of production, the intent of sale, and the marketing claims made about the product.
The Act acknowledges that many Rongoā Māori products incorporate indigenous flora and traditional preparation methods. The regulations will need to provide clear guidance on when these products transition from being part of an exempt traditional practice to becoming a regulated therapeutic product. This is critical for practitioners who may wish to share their knowledge more broadly, or for enterprises that seek to develop and market products based on Rongoā principles. The ongoing consultation and development of secondary legislation (regulations) will be crucial in drawing these lines with clarity and cultural sensitivity. It will require careful consideration of intellectual property rights related to traditional knowledge, benefit-sharing, and ensuring that any commercialisation does not undermine the integrity or authenticity of Rongoā Māori. The aim is to create a pathway for innovation and accessibility while protecting the cultural heritage and practices that define Rongoā.

Compliance Requirements for Commercial Product Makers
While traditional Rongoā Māori practices are intended to receive exemptions, the Therapeutic Products Act imposes stringent compliance requirements on commercial product makers, including those who produce Rongoā-derived or Rongoā-inspired products for sale to the general public. This distinction is crucial: if a product is manufactured, marketed, and sold with therapeutic claims, it will generally be treated like any other therapeutic product under the new regulatory regime, regardless of its traditional origins. The Act aims to ensure that all commercially available therapeutic products meet consistent standards of safety, quality, and efficacy, protecting consumers while fostering a level playing field for manufacturers.
Commercial product makers will need to navigate a series of new obligations, starting with product authorisation. Before a therapeutic product can be supplied in New Zealand, it will need to be authorised by the new regulator. This authorisation process involves submitting comprehensive data demonstrating the product’s quality, safety, and intended therapeutic benefits. For products incorporating Rongoā elements, this could pose unique challenges, as traditional knowledge often relies on centuries of empirical observation rather than modern randomised controlled trials. The regulator will need to establish pathways that are sensitive to different forms of evidence, potentially including traditional use evidence, while still ensuring a robust assessment of safety and efficacy.
Manufacturing Standards and Quality Control
Beyond product authorisation, commercial manufacturers will be subject to strict manufacturing and quality control standards. The Act introduces requirements for Good Manufacturing Practice (GMP), which mandate robust quality management systems, validated manufacturing processes, and rigorous testing of raw materials and finished products. This includes facilities, equipment, personnel, and documentation. For smaller enterprises or those transitioning from traditional preparation methods to commercial production, achieving GMP compliance can be a significant undertaking, requiring investment in infrastructure, training, and quality assurance personnel. The goal is to prevent contamination, ensure consistency, and guarantee that each batch of product meets its specifications. This is a non-negotiable aspect of the new regime, designed to safeguard public health by ensuring product integrity from source to consumer.
Furthermore, commercial entities will be responsible for pharmacovigilance and post-market surveillance. This involves monitoring the product’s safety profile once it is on the market, collecting and reporting adverse events, and taking corrective actions if safety concerns arise. This continuous oversight ensures that any unforeseen risks are identified and managed promptly. For products with a long history of traditional use, this requirement will necessitate establishing systematic methods for collecting and evaluating safety data in a modern context. It also places a burden on manufacturers to have robust systems in place for traceability, recall, and communication with both the regulator and the public in case of product issues.
Navigating the Regulatory Landscape for Rongoā-Based Products
For commercial makers of Rongoā-based products, the path to compliance will require strategic planning and a proactive approach. This includes understanding the specific classifications their products fall under, engaging early with the regulator, and potentially seeking expert advice on regulatory affairs, quality assurance, and clinical evidence. It may involve investing in research to scientifically validate traditional claims, or adapting manufacturing processes to meet modern GMP standards without compromising the authenticity of the Rongoā components. The Act offers flexibility for regulations to address specific product types, suggesting that tailored pathways might emerge for traditional and natural health products. However, the onus will be on commercial entities to demonstrate their products meet the required standards for safety and quality. This shift requires a deep understanding of both Rongoā principles and modern regulatory science, bridging two distinct knowledge systems to achieve compliance and ensure consumer confidence.

Navigating Claims and Labeling Laws
The Therapeutic Products Act introduces a robust framework for claims and labeling, significantly impacting how therapeutic products, including those with Rongoā origins, can be advertised and presented to the public. The fundamental principle is that all claims made about a therapeutic product – whether on its label, in advertising, or other promotional materials – must be accurate, truthful, balanced, and substantiated by evidence. This is a critical consumer protection measure designed to prevent misleading or exaggerated claims and ensure that consumers can make informed decisions about their health choices. For products derived from traditional knowledge, this presents a unique challenge: how to communicate traditional understanding and benefits within a regulatory environment that often prioritises scientific, Western-style evidence.
Under the new Act, the regulator will have powers to approve or review claims, and to issue directions regarding advertising content. Manufacturers and suppliers will be responsible for ensuring that all promotional material, including websites, social media, and packaging, complies with the advertising standards and prohibitions. This includes explicit restrictions on making false or misleading statements, unsubstantiated claims, or claims that suggest a product can cure serious diseases unless such claims are explicitly authorised and backed by robust scientific evidence. The goal is to create a transparent and trustworthy marketplace for therapeutic products, where consumers can rely on the information provided.
Balancing Traditional Knowledge with Evidentiary Standards
For Rongoā-based products, navigating these claims and labeling laws requires a delicate balance. Traditional Rongoā knowledge often includes statements about spiritual, emotional, and physical well-being that may not easily translate into quantifiable scientific endpoints. The challenge lies in respecting and reflecting these traditional understandings while also adhering to the Act’s requirements for evidence-based claims. There is an opportunity for the regulator, in consultation with the Rongoā Māori advisory body, to develop specific guidelines that accommodate traditional evidence and ways of knowing. This might involve recognising a different standard of evidence for traditional use, provided that safety and quality are assured, or developing specific categories of claims that are culturally appropriate and informative without being misleading in a Western scientific context.
Labeling requirements will also be comprehensive, mandating the inclusion of essential information such as ingredients, dosage instructions (if applicable), batch numbers, expiry dates, and warnings or contraindications. For products derived from Rongoā rākau, clear identification of the plant species used, its origin, and any specific traditional preparation methods that impact its use will be crucial. The Act aims to provide consumers with all necessary information to safely and effectively use a product. Furthermore, the use of Te Reo Māori on labels and in advertising will need to be considered carefully to ensure accuracy and cultural appropriateness, while also meeting the regulatory requirements for clarity and consumer understanding.
Avoiding Misleading Advertising and Unsubstantiated Claims
The consequences of non-compliance with claims and labeling laws can be significant, ranging from formal warnings and product recalls to substantial fines and prosecution. The regulator will have powers to investigate complaints, issue infringement notices, and impose sanctions. This places a strong onus on commercial product makers to be meticulous in their communication and marketing strategies. For those involved in the commercialisation of Rongoā-based products, this means moving beyond anecdotal evidence to compile verifiable data, even if it is traditional use evidence presented in a structured manner. It also necessitates a clear understanding of what constitutes a ‘therapeutic claim’ versus a ‘general health claim’ or a ‘traditional use statement,’ as each may have different evidentiary thresholds. Engagement with regulatory experts and the Rongoā Māori advisory body will be essential to ensure that the spirit of Rongoā is upheld while navigating the strictures of modern consumer protection law.

Conclusion: Charting a Course for Rongoā in a Regulated Future
The Therapeutic Products Act represents a pivotal moment for health products in Aotearoa New Zealand, including the evolving landscape of Rongoā Māori. Its enactment underscores a national commitment to public health and safety, introducing a robust, lifecycle-based regulatory framework for therapeutic products. For Rongoā, the Act presents a dual reality: a profound recognition of its cultural significance and a clear pathway for traditional practices to be largely exempted from the Act’s commercial requirements, juxtaposed with stringent compliance demands for any commercially manufactured Rongoā-derived products. Navigating this new terrain will require careful consideration, collaboration, and a deep understanding of both traditional knowledge systems and contemporary regulatory science.
The successful implementation of the Act, particularly concerning Rongoā Māori, hinges on the continued, meaningful engagement with the Rongoā community. The establishment of a Rongoā Māori advisory body is a crucial step towards ensuring that regulations are culturally appropriate, informed by tikanga, and effective in protecting both traditional practices and public safety. This collaborative approach is essential for developing clear guidelines on exemptions, defining the boundaries between traditional practice and commercialisation, and establishing culturally sensitive evidentiary standards for claims and labeling.
For traditional practitioners, the Act offers reassurance that their customary healing practices will be largely protected, reinforcing the mana (prestige, authority) of Rongoā. For commercial product makers, it necessitates a paradigm shift towards rigorous quality management, scientific substantiation, and transparent communication. The challenge lies in translating traditional efficacy and safety knowledge into a framework that meets modern regulatory expectations without diluting the essence of Rongoā. The journey forward will undoubtedly be complex, but with a commitment to bicultural partnership and mutual understanding, the Therapeutic Products Act has the potential to safeguard Rongoā Māori as a taonga, while ensuring that all therapeutic products available to New Zealanders are safe, effective, and of high quality.
People Also Ask
What is the primary purpose of the Therapeutic Products Act?
The Therapeutic Products Act aims to ensure the safety, quality, and efficacy of all therapeutic products available in New Zealand, replacing the outdated Medicines Act 1981 with a comprehensive, lifecycle-based regulatory system to protect public health.
How does the new Act impact traditional Rongoā Māori practices?
The Act includes provisions to largely exempt traditional Rongoā Māori practices, such as karakia, mirimiri, and customary use of rongoā rākau, when conducted by traditional practitioners within a customary context, acknowledging their unique cultural status.
Will commercial products based on Rongoā Māori be regulated under the Act?
Yes, commercially manufactured and sold therapeutic products that incorporate Rongoā elements or make therapeutic claims will generally be subject to the Act’s full regulatory requirements, including product authorisation, manufacturing standards, and advertising rules.
What kind of exemptions are available for Rongoā practitioners?
The Act allows for specific regulations to be made that exempt certain products or practices from its scope. A Rongoā Māori advisory body will help define these exemptions to protect traditional healing without over-regulation.
What are the key compliance requirements for commercial Rongoā product makers?
Commercial makers must obtain product authorisation, adhere to Good Manufacturing Practice (GMP) standards for quality and safety, and comply with strict rules regarding claims, labeling, and post-market surveillance for their products.
How will the Act balance traditional Rongoā claims with modern evidentiary standards?
The Act requires claims to be substantiated. For Rongoā-based products, the regulator, guided by the Rongoā Māori advisory body, will need to explore pathways that respectfully incorporate traditional use evidence while ensuring public safety and preventing misleading information.